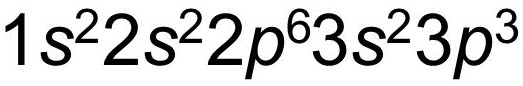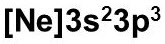Create your Eureka Connections Page for Unit 2
The Eureka! Connections pages are located at the beginning of each new unit in your Interactive Science Notebook
- The next two side-by-side pages in your notebook.
- In the center (or close to it) write your big idea or problem statement for the unit. (Hint: look at the heading of this page)
- After each activity in the unit, write a statement on this page spread which conveys the concept learned.
- Share with lab partners and other classmates. Make connections between the content (Conceptually in your mind and Visually on the page with arrows.)
- Use these "lines of evidence" and their connections to write your Eureka! Thesis at the end of the unit.
(All assignments are due the the following class meeting.)
Chapter 3: Atoms - The Building Blocks of Matter
|
|
The Atom: From Philosophical Idea to Scientific TheoryCovers the history and development of atomic theory, from Democtritus to Dalton to the modern era.
Tuesday (10/4): Read pages 63-66 of our class textbook ("Modern Chemistry"). Watch the video "The Nucleus." Left-side work: Explain the Law of Conservation of Mass, Law of Definite Proportions, Law of Multiple Proportions. Summarize the 5 points of Dalton's Atomic Theory. Identify the relation ship between Dalton's Theory and the three laws mentioned above. Update the Eureka! Connections page for Unit 2.
The Structure of the AtomCovers the experiments that led to the discovery of the electron and the atomic nucleus - and the principle properties of these subatomic particles.
Wednesday (10/5): Read pages 68-72 of our class textbook ("Modern Chemistry"). Left-side work: No homework for this day.
Friday (10/7): Watch the video "The Creation of Chemistry." .Left-side work: Complete questions 1, 2, and 5 on page 72 of our class textbook ("Modern Chemistry"). Update the Eureka! Connections page for Unit 2.
Counting AtomsOutlines the manner in which the number of atoms of an element and the number of an atom's subatomic particles can be expressed and measured.
Tuesday (10/11): Read and do the practice problems on pages 73-88 of our class textbook ("Modern Chemistry"). Left-side work: Complete the Counting Protons, Electrons, & Neutrons" worksheet and the "Calculating Average Atomic Mass" worksheet. Update the Eureka! Connections page for Unit 2.
Wednesday (10/12): Left-side work: Complete the analysis and conclusion portion of the "Conservation of Mass" Lab. Update the Eureka! Connections page for Unit 2.
Friday (10/14): Read and do the practice problems on pages 79-83 of our class textbook ("Modern Chemistry"). Left-side work: Complete the "Average Atomic Mass" worksheet and the "Grams, Moles, & Number of Atoms" worksheet that were handed out in class. Update the Eureka! Connections page for Unit 2.
Tuesday (10/18): Left-side work: Complete Grams, Moles, and Molecules Practice sheet. Update the Eureka! Connections page for Unit 2.
Wednesday (10/19): Left-side work: Complete Study Guide for Chapter 3. Update the Eureka! Connections page for Unit 2.
Friday (10/21): Left-side work: Complete Study Guide for Chapter 3. Update the Eureka! Connections page for Unit 2.
|
Chapter 4: Arrangement of Electrons in Matter
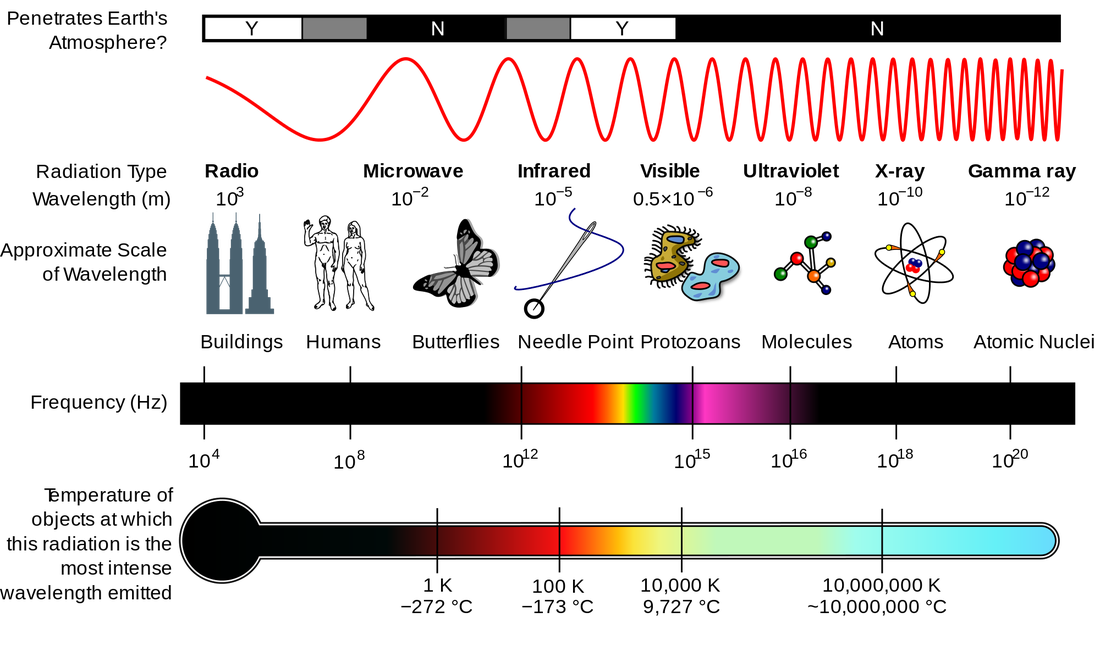
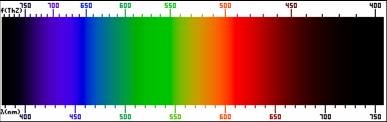
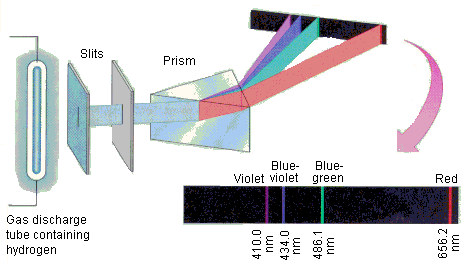
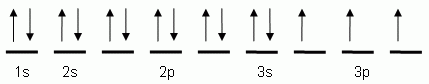
Electromagnetic SpectrumContinuous SpectrumHydrogen Line-emission SpectrumOrder of Filling Orbitals with ElectronsOrbital Notation Example - Phosphorus (15):Electron Configuration Notation Example - Phosphorus (15):Noble-Gas Notation Example - Phosphorus (15): |
The Development of a New Atomic ModelDescribes the principles of electromagnetic radiation and the development of Bohr's model of the atom.
Tuesday (10/25): No Left-side work: Update the Eureka! Connections page for Unit 2. Watch the videos: Photoelectric Effect Demonstration and The Original Double Slit Experiment.
Thursday (10/27): Read pp.91-97 in Modern Chemistry. Left-side work: Answer Page 97: 1-8. Update the Eureka! Connections page for Unit 2. Study the images to the left: "Electromagnetic Spectrum," "Continuous Spectrum," and "Hydrogen Line-emission Spectrum. Watch the videos: The Electromagnetic Spectrum and How to Make a Spectroscope.
The Quantum Model of the AtomDescribes the location of electrons around the nucleus from a wave-mechanical (quantum) perspective using quantum numbers.
Tuesday (11/1): Read pp.98-104 in Modern Chemistry. Left-side work: Answer Page 104: 1-3. Update the Eureka! Connections page for Unit 2. Watch the videos: What is the Heisenberg Uncertainty Principle?, Quantum Mechanics: Schrödinger's Discovery of the Shape of Atoms, and Is This What Quantum Mechanics Looks Like?.
Electron ConfigurationsDiscusses the rules used to determine the electron configurations of the elements and introduces electron configuration notations.
Wednesday (11/3): Read pp.105-106 in Modern Chemistry. Left-side work: Complete the "Orbital Notations" worksheet. Update the Eureka! Connections page for Unit 2. Review images to the left, entitled: "Order of Filling Orbitals with Electrons" and "Orbital Notation Example - Phosphorus (15)."
Thursday (11/4): Read pp.107 & 110. Left-side work: Complete the "Electron-Configuration Notation" worksheet. Update the Eureka! Connections page for Unit 2.
Tuesday (11/8): Read pp.108-109 & 111-116 in Modern Chemistry. Watch the video: The Electron: Crash Course Chemistry #5. Left-side work: Complete the "Noble-Gas Notation" worksheet. Update the Eureka! Connections page for Unit 2.
Wednesday (11/9): Left-side work: Complete the "3 Styles of Electron Configuration Practice" worksheet.
Tuesday (11/15): Left-side work: Complete the "Chapter 4 Study Guide.". Review for the test - which will be available on Schoology Wednesday - Sunday (16 Nov 2016 - 20 Nov 2016).
Wednesday -Sunday (11/16 - 11/20): Chapter Work & Test Due: Take Chapter 4 test on Schoology (it will be timed, so once you start, you cannot stop - there are no re-takes). Be sure to submit your notebook for a complete update of grades by Monday (11/21).
|
Chapter 5: The Periodic Law
|
History of the Periodic TableCovers the work of Mendeleev (and other chemists) in developing the periodic table and explains how the periodic law is used to predict elements' physical and chemical properties.
Friday (11/18): Watch the video: The Periodic Table: Crash Course Chemistry #4. Left-side work: Complete the "Periodic Table Coloring Activity" worksheet. Update the Eureka! Connections page for Unit 2.
Tuesday (11/22): Left-side work: Modern Chemistry pg. 129: 1-4. Update the Eureka! Connections page for Unit 2. Read The Radioactive Boyscout...if you are interested.
Electron Configuration and the Periodic TableExplains the relationship between electron configuration and the arrangement of elements in groups, blocks, and periods of the periodic table, as well as the elements' general properties.
Wednesday (11/23): Left-side work: Modern Chemistry pg. 141: 1-6. Update the Eureka! Connections page for Unit 2.
Electron Configuration and Periodic PropertiesFurther explores the relationship between the periodic law and electron configuration, including trends in the properties of electron affinity, electronegativity, ionization energy, atomic radii, and ionic radii.
Tuesday (11/29): Left-side work: Complete "Periodic Table Trends Worksheet," available on the class Schoology page.. Update the Eureka! Connections page for Unit 2.
Wednesday -Sunday (11/30 - 12/11): Chapter Work & Test Due: Take Chapter 5 test on Schoology (it will be timed, so once you start, you cannot stop - there are no re-takes).
| ||||||
Chapter 21: Nuclear Chemistry
|
|
The Nucleus
Tuesday (12/6): Watch the video: Nuclear Chemistry: Crash Course Chemistry #38. Left-side work: Modern Chemistry pg. 644: 2-4. Update the Eureka! Connections page for Unit 2.
Radioactive DecayOutlines different types of radioactive decay and demonstrates calculations involving the half-life of an isotope.
Nuclear RadiationSurveys the ability of nuclear radiation to penetrate materials, methods of detecting radiations, and other applications of nuclear radiation.
Nuclear Fission and Nuclear FusionExplains the difference between fission and fusion and their potential as energy sources.
|